Research
Please visit the above links to have a more detailed look at our research projects. Below is a list of Centers that we are leading or affiliated with.
NSF MBM at UIUC - Miniature Brain Machinery
NSF STC EBICS at MIT/GT/UIUC - Emergent Behavior of Integrated Cellular Systems
NSF IGERT at UIUC - Cellular and Molecular Mechanics and Bionanotechnology
(download brochure)NIH Training Grant at UIUC - Midwestern Cancer Nanotechnology Training Center
(download brochure)NSF CiiT (I/UCRC) at UIUC - Center for Innovative Instrumentation Technology
NSF NSEC at OSU - Center for Affordable Nanoengineering for Polymeric Micro and Nanodevices
Research:
Characterization of single cell mass response
A complex relationship exists between the behavior of a cell and its physical properties. There is strong interest in understanding the mechanisms through which the physical proper- ties of a cell influence growth, differentiation, cell cycle progression, and apoptosis. Nanobiomechanics, which is the measuring of material properties at the smallest length scale, is an emerging area of research that has the ability to evolve the study of human disease. This field has helped elucidate mechanisms of disease progression and provided additional tools in the fight against disease.
Much of the research on cell growth focuses on the temporal dynamics of growth rate. Variations in growth rate over the cell cycle may elucidate mechanisms underlying cell growth better than the magnitude of growth rate alone. There are two major models used to analyze the cell cycle: one based on an exponential increase and another based on a linear increase. Exponential growth rate for an individual cell is proportional to cell size mass, volume, or density during the cell cycle. Linear growth rate for an individual cell is constant meaning the cell increases size by the same amount regardless of its current size or state.
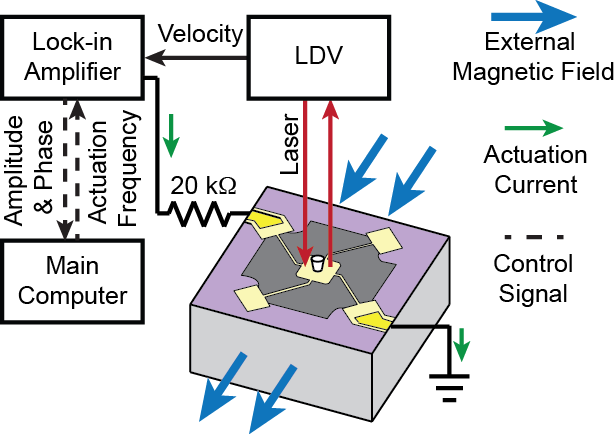
We developed an improved MEMS resonator sensor that can be used to directly measure the biophysical properties, mass, and growth rate of single adherent cells. These devices detect mass through resonant shift, and are able to be operated in gaseous or aqueous environment. In order to overcome damping from surrounding fluid these devices must be driven magnetically to achieve a distinguishable resonance peak. However, our measurement technique offers a combination of complex elastic and viscoelastic dynamic properties of cells. Decoupling the relationship between the cell’s dynamics and the apparent mass reported by the sensor is of utmost importance. Understanding this relationship will further empower the measurement technique, enabling even more prudent investigations that will benefit efforts in cancer diagnosis and treatment, biological accurate design, cell-to-cell interfacing, and tissue engineering, among others.